Welcome to SittuAI Your Regulatory Submission Copilot
Harness the power of advanced AI to streamline your 510(k) submissions. Our intelligent platform automates 80% of your regulatory documentation while ensuring FDA compliance.
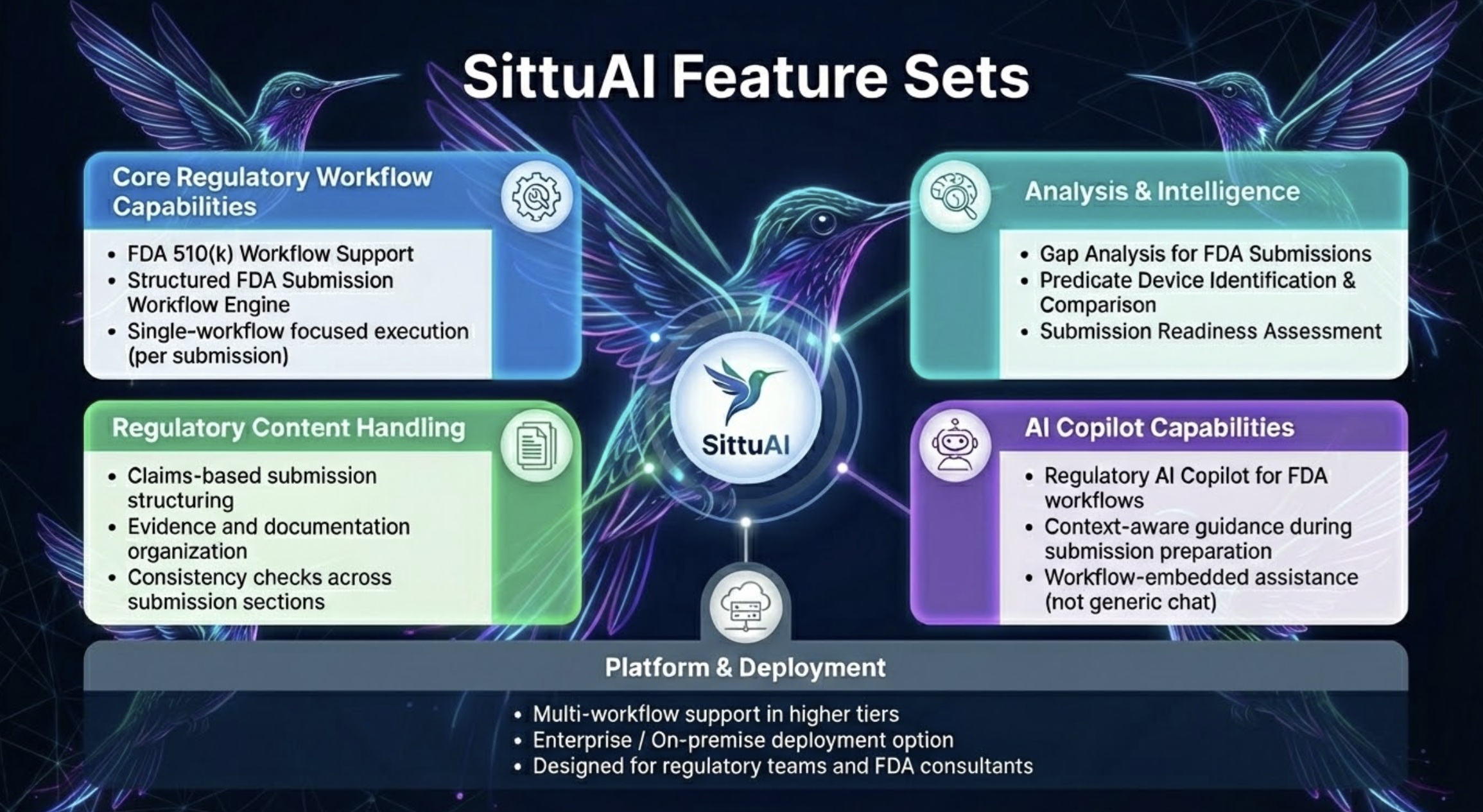
Intelligent Capabilities
Powered by proprietary AI technology designed specifically for FDA regulatory workflows
Predicate Intelligence
Instantly finds and analyzes similar cleared devices from our comprehensive FDA database.
Copilot Chat
Automatically drafts 24+ regulatory documents using your device profile and FDA requirements.
AI Document Generation
Automatically generates 24+ compliant regulatory documents tailored to your device profile and FDA requirements.
Knowledge Graph
Connected intelligence that understands device relationships, regulations, and FDA guidance documents.
Compliance Validation
Real-time validation against 21 CFR requirements and current FDA guidance documents.
Simple 4-Step Process
From device profile to FDA-ready submission package
Define Your Device
7-step guided wizard
AI Analysis
Find predicates & gaps
Generate Documents
24+ docs auto-drafted
Export Package
eSTAR-ready format
From Device Profile to FDA-Ready Submission
A purpose-built platform that guides medical device companies through every step of the 510(k) journey — intelligently, efficiently, and with confidence.
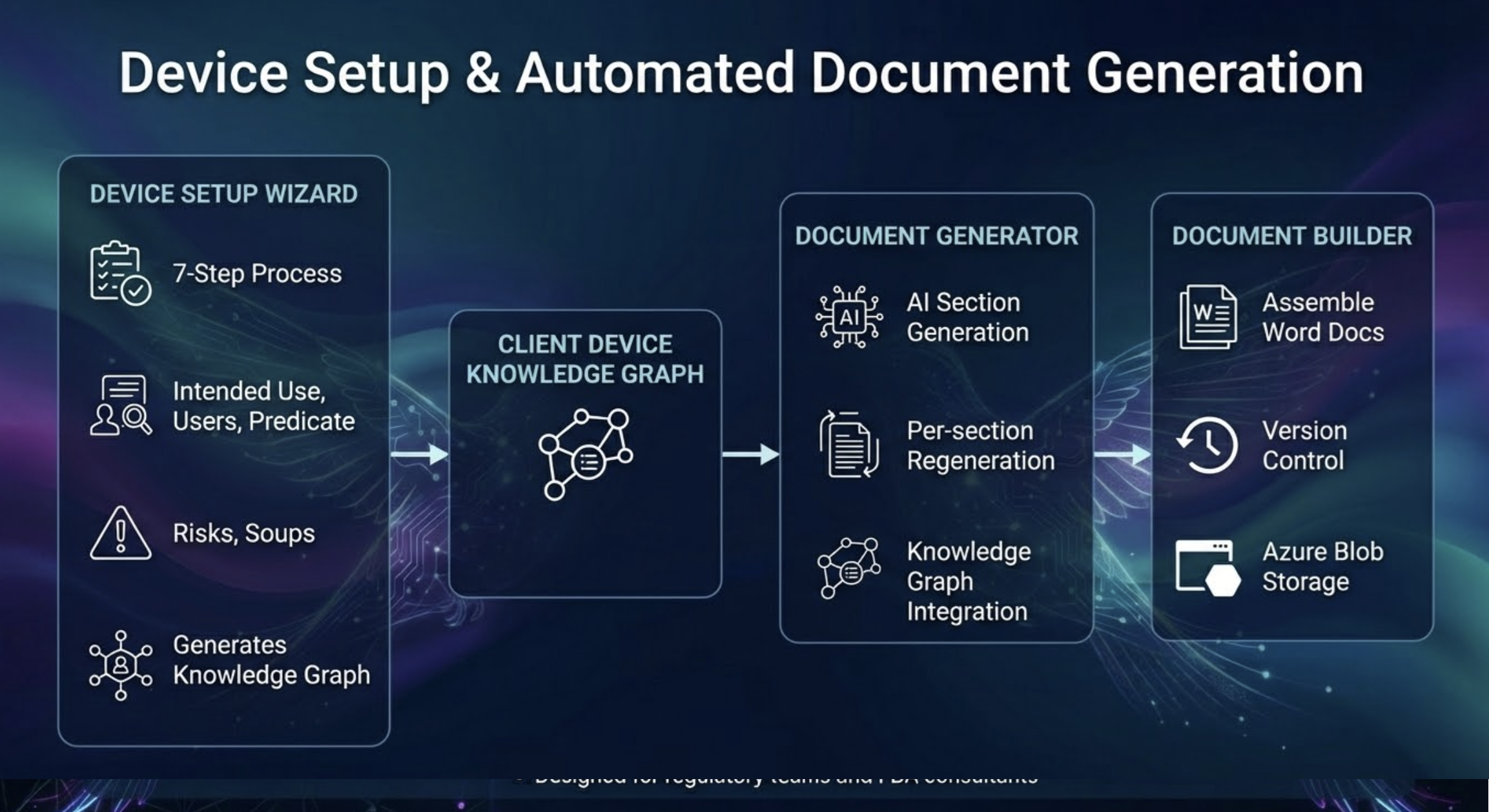
Intelligent Device Setup
Get started in minutes with our guided 7-step device onboarding wizard. Define your device's intended use, classification, target users, and predicate strategy. Our AI builds a rich Device Knowledge Graph that powers every downstream workflow — from document generation to predicate matching.

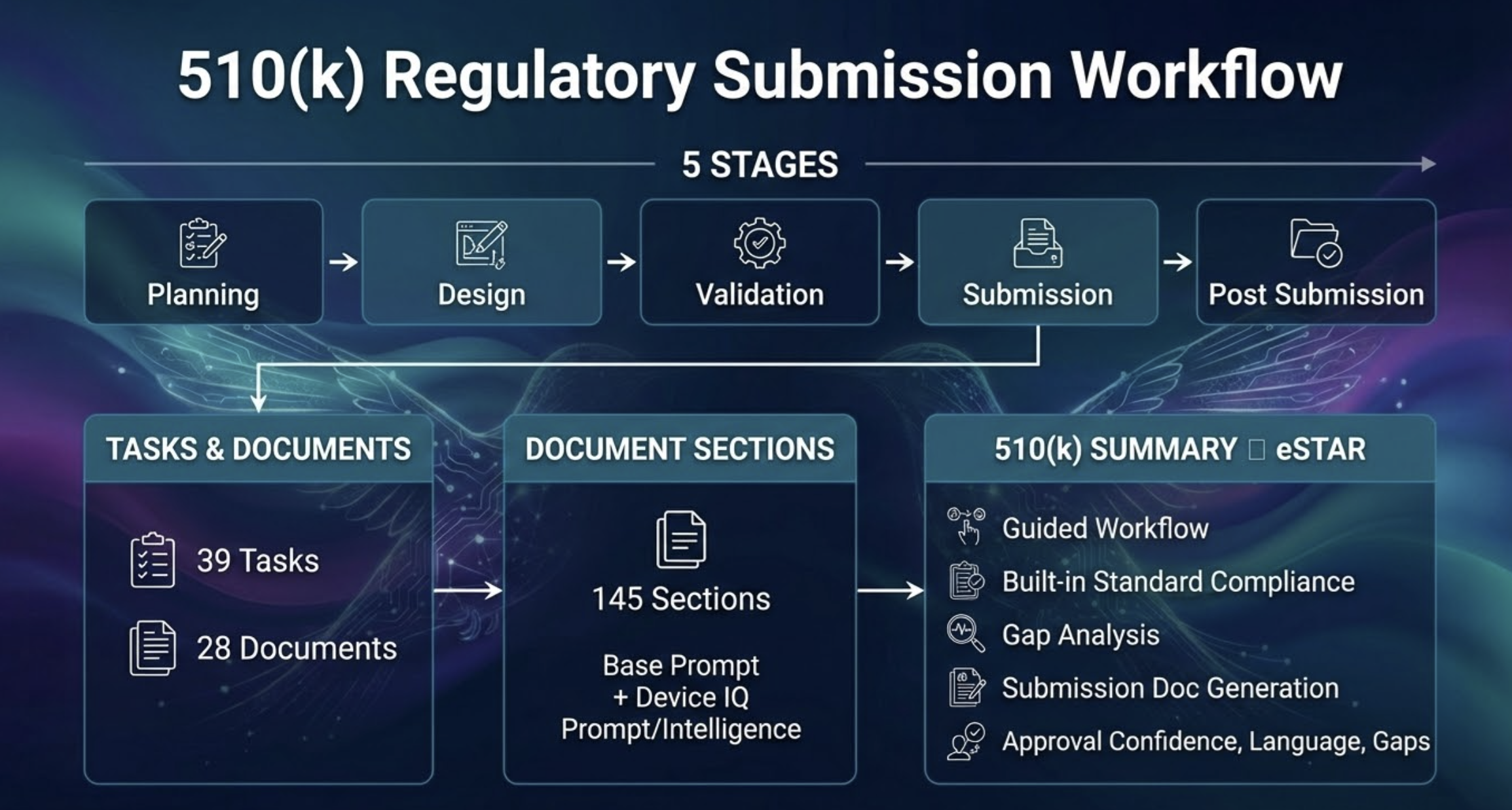
Structured Regulatory Workflow
Follow a proven, stage-gated submission workflow built around FDA expectations. SittuAI breaks the 510(k) journey into clear phases so your team always knows what's next, what's pending, and what's complete — with full progress visibility across every stage.

AI-Powered Document Generation
Generate all 24+ regulatory documents from a single device profile. Each document comes with section-level tracking, individual approval workflows, and AI-assisted content generation per section — so your team can review, edit, and approve with precision rather than starting from scratch.

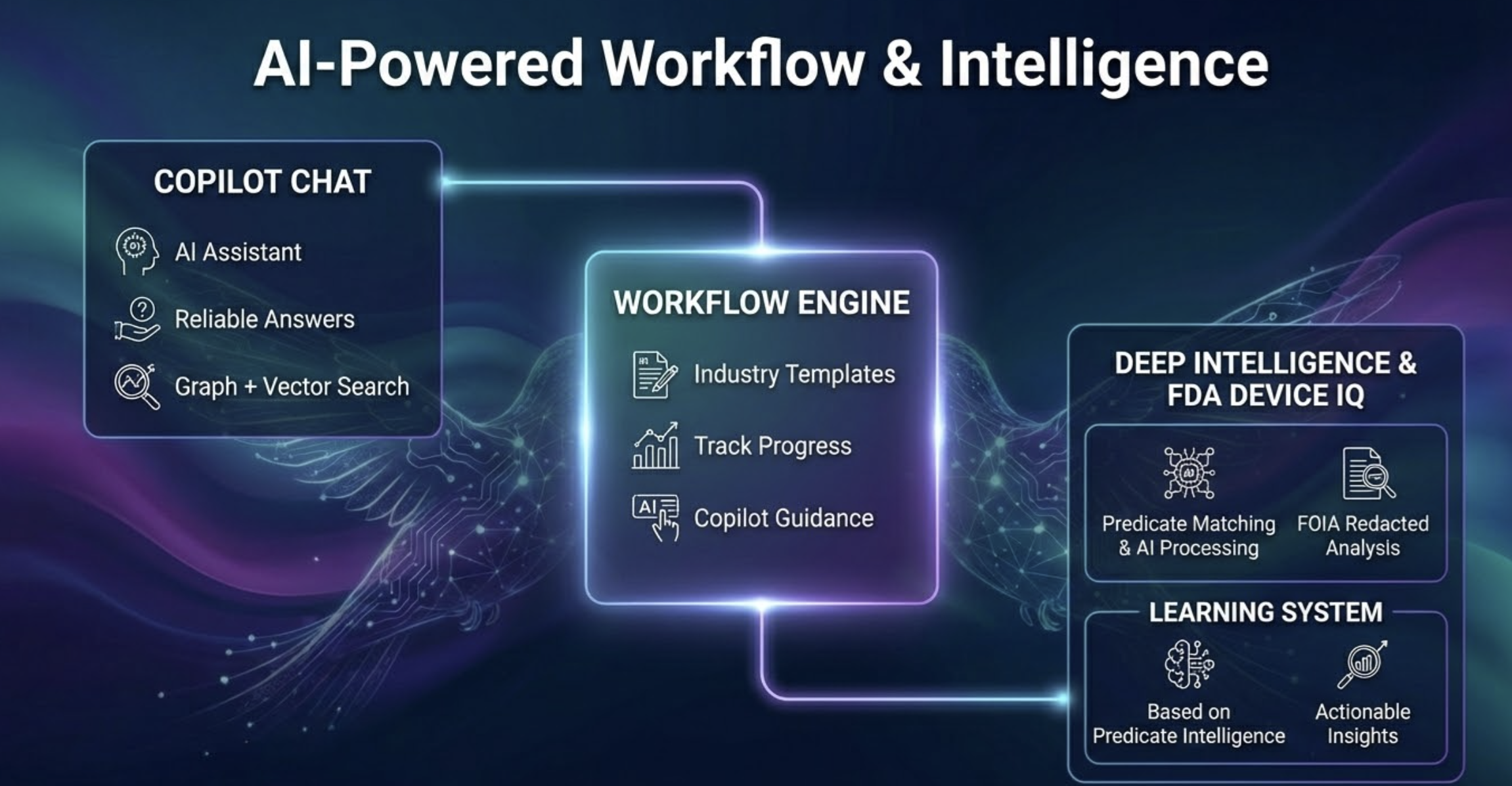
Regulatory Copilot Chat
Ask anything. Get precise, grounded answers. Our Copilot is trained on FDA guidance documents, 510(k) precedents, and your device's own knowledge graph — delivering consistent, reliable answers specific to your submission rather than generic AI responses.

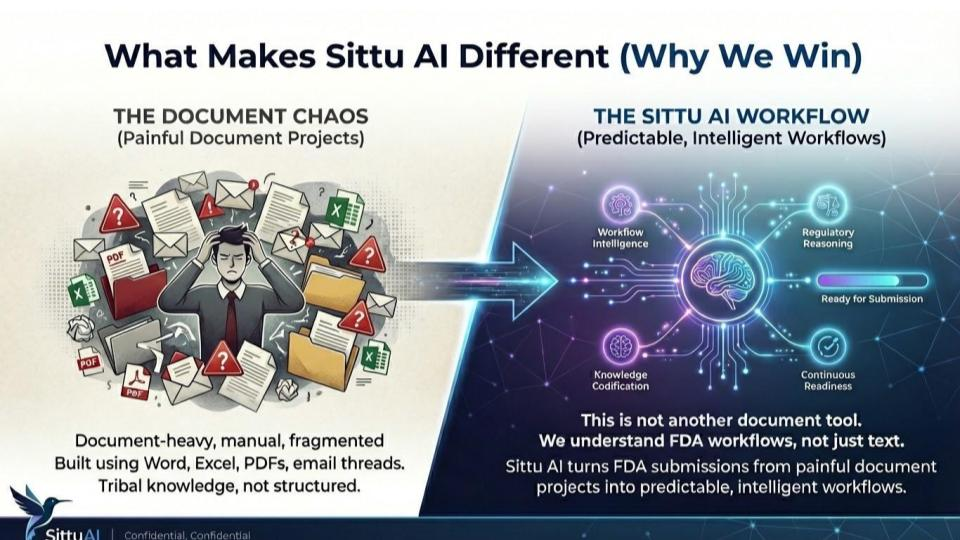
The SittuAI Advantage
Unlike generic AI tools, SittuAI is purpose-built for FDA 510(k) submissions. Every feature — from predicate intelligence to document assembly — is designed around real regulatory workflows. The result: up to 60% faster submission preparation, fewer revision cycles, and teams that spend time on strategy, not paperwork.

Ready to Transform Your 510(k) Submission?
Join medical device companies who are submitting faster, smarter, and with confidence.
Grow your consulting practice — without growing the grind.
80% of your time is spent on documentation, not expertise. SittuAI handles the drafting work so you can focus on what clients actually pay you for: validation, strategy, and FDA know-how. Join our platform for free and get matched with medical device companies who need your expertise.
Serve more clients, faster
AI drafts the documents needed per submission. You validate, not rewrite. Take on 3-5× more clients in the same time.
Get matched with new customers
Medical device, Pharma & Cosmetic companies and SaMD startups come to us for help. We route qualified clients directly to consultants on our platform.
Free to join.
No subscription, no platform fees. Use SittuAI's AI-powered Predicate Matching & Co Pilot chat for your own client engagements.
Cut submission time in half
Validated by a working FDA consultant: 60% reduction in total 510(k) submission time. Deliver faster, impress clients, win more repeat business.
Your expertise stays yours
You validate every document. Your name, your credentials, your sign-off. SittuAI is the leverage — you're still the expert.
510(k), De Novo, PMA ready
Pre-built workflow templates for every major FDA submission pathway. Knowledge graph built from FDA regulations, guidance, and real precedents.
Takes 2 minutes. We'll be in touch within 1 business day.
Latest FDA Regulatory Insights
Expert-written guides on 510(k), De Novo, PMA submissions, and compliance strategy.
Classification of Digital Therapeutics: A Practical Guide to Determining Your FDA Regulatory Pathway
Digital therapeutics present a complex FDA classification challenge because software treatments don't fit traditional device categories. Choosing the correct classification pathway—Class I, II, or III—determines your premarket strategy, timeline, and budget, making it the most consequential early-stage regulatory decision.
FDA Software as a Medical Device (SaMD): A Practical Interpretive Guide
**FDA regulates Software as a Medical Device (SaMD)—software performing medical functions independently on general-purpose platforms like smartphones or cloud servers—under a risk-based framework, with exclusions for administrative tools, wellness apps, and clinical decision support meeting strict criteria. Companies must navigate multiple regulatory instruments including FDA's 2019 AI/ML guidance and 2023 predetermined change control plan guidance to determine premarket clearance requirements.**
In Vitro Diagnostic Device Classification: A Practical Guide to Getting It Right
**Summary:** Correct IVD classification determines your regulatory pathway—a Class I exemption takes weeks, Class II 510(k) takes 6–12 months, and Class III PMA takes 18–36 months and millions in costs. The FDA classifies IVDs under 21 CFR Parts 862–892 based on risk level, with each device assigned a product code and class that dictates required evidence, controls, and post-market obligations.
Ready to Transform Your Regulatory Submissions?
Join leading medical device companies who have reduced their 510(k) preparation time by 60%.